Compounds containing just carbons and hydrogens are the most basic compounds encountered in organic chemistry. These compounds are referred to as hydrocarbons. On the most basic level, hydrocarbons can be divided into three groups: those containing just single bonds, those containing one or more double bonds, and those containing one or more triple bonds. Before discussing how to name these compounds, it is instructive to examine how they are represented by chemists.
Table of contents:
- Drawing Hydrocarbons
- Nomenclature:
Drawing Hydrocarbons
Recall that when carbon makes four bonds, it adopts the tetrahedral geometry. In the tetrahedral geometry, only two bonds can occupy a plane simultaneously. The other two bonds point in back or in front of the plane. In order to represent the tetrahedral geometry in two dimensions, solid wedges are used to represent bonds pointing out of the plane of the drawing toward the viewer, and dashed wedges are used to represent bonds pointing out of the plane of the drawing away from the viewer. Consider the following representation of the molecule methane:
 |
| Two dimensional representation of methane |
In the above drawing, the two hydrogens connected by solid lines, as well as the carbon in the center of the molecule, exist in a plane (specifically, the plane of the computer monitor / piece of paper, etc.). The hydrogen connected by a solid wedge points out of this plane toward the viewer, and the hydrogen connected by the dashed wedge points behind this plane and away from the viewer.
In drawing hydrocarbons, it can be time-consuming to write out each atom and bond individually. In organic chemistry, hydrocarbons can be represented in a shorthand notation called a skeletal structure. In a skeletal structure, only the bonds between carbon atoms are represented. Individual carbon and hydrogen atoms are not drawn, and bonds to hydrogen are not drawn. In the case that the molecule contains just single bonds (sp3 bonds), these bonds are drawn in a "zig-zag" fashion. This is because in the tetrahedral geometry all bonds point as far away from each other as possible, and the structure is not linear. Consider the following representations of the molecule propane:
 |
 |
| Full structure of propane | Skeletal structure of propane |
Only the bonds between carbons have been drawn, and these have been drawn in a "zig-zag" manner. Note that there is no evidence of hydrogens in a skeletal structure. Since, in the absence of double or triple bonds, carbon makes four bonds total, the presence of hydrogens is implicit. Whenever an insufficient number of bonds to a carbon atom are specified in the structure, it is assumed that the rest of the bonds are made to hydrogens. For example, if the carbon atom makes only one explicit bond, there are three hydrogens implicitly attached to it. If it makes two explicit bonds, there are two hydrogens implicitly attached, etc. Note also that two lines are sufficient to represent three carbon atoms. It is the bonds only that are being drawn out, and it is understood that there are carbon atoms (with three hydrogens attached!) at the terminal ends of the structure.
Alkane nomenclature
When hydrocarbons contain only single bonds, they are called alkanes. Alkanes are named using a prefix for the number of carbon atoms they contain, followed by the suffix -ane. The following table lists the rules for naming alkanes.
| Number of carbon atoms |
Prefix | Compound name | Image |
|---|---|---|---|
| 1 | Meth- | Methane |  |
| 2 | Eth- | Ethane |  |
| 3 | Prop- | Propane |  |
| 4 | But- | Butane |  |
| 5 | Pent- | Pentane |  |
| 6 | Hex- | Hexane | 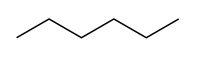 |
| 7 | Hept- | Heptane |  |
| 8 | Oct- | Octane |  |
| 9 | Non- | Nonane |  |
Alkane nomenclature is straightforward; the only difficulties come if one of the hydrogen or carbon atoms on the molecule is replaced by another atom or group. When this takes place, the group which replaces the hydrogen or carbon is called a substituent.
Since the only kind of molecules we've discussed so far are alkanes, let's consider the situation in which one of the hydrogen atoms on an alkane has been replaced by another alkane. Consider the following molecule, 3-methypentane:
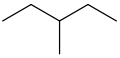 |
| Skeletal structure of 3-methylpentane |
Consider the long chain of five carbon atoms at the top of the image. If this were all that composed the molecule, it would simply be called pentane. However, one of the hydrogens on the carbon third from the end has been replaced with an alkane, specifically methane. How are we to name this molecule?
- First, we identify the longest chain of carbon atoms. We name this alkane. It will
serve as the root name for the molecule.
- In the example above, the root name is pentane.
- Next, we number the carbon atoms, starting at the end that gives the substituent the lowest number.
- In the example above, we can count from either end and arrive at 3 for the substituent.
- Next, we name the substituent as if it were an independent alkane. However, we replace suffix -ane with -yl. This name will serve as the prefix.
- In the example above, methane is the substituent, so we call it methyl.
- The compound is named "number-prefixrootname".
- In the example above, the name is 3-methylpentane
Let's try some more complicated examples. What happens if the alkane has more than one substituent? In this case, the rules above are followed, and the carbons on the longest chain are numbered to give the lowest number possible to one of the substituent. The substitients are then all named in the prefix (e.g. 2-ethyl,3-methyl). If more than one substituent is attached to the same carbon atom, the number of that carbon atom is repeated to indicate the number of substituents and the prefixes di- (2) or tri- (3) are used. If there are more than one substituent on different carbon atoms, the prefixes are ordered alphabetically (e.g. ethene before methane). The prefixes di- and tri- are ignored when considering alphabetical order. Consider the following compound:
 |
| Skeletal structure of 4-ethyl-2,2-dimethylheptane |
The longest carbon chain has seven carbon atoms, so the root name is heptane. Numbering from the right gives the lowest number to the first substituent. There are two methyl substituents at the second carbon atom, so we use the prefix 2,2-dimethyl. There is another substituent on the fourth carbon atom, so we use the prefix ethyl. Ethyl comes before methyl alphabetically, so we name the compound 4-ethyl-2,2-dimethylheptane.
Let's consider one final example, when the substituent is not a linear alkane. If the substituent is branched, we consider these branches to be substituents of the original substituent (confused yet?). The substituent's substituents must be named using the rules outline above. The final name of the original substituent is placed in parentheses. Consider the following molecule:
 |
| Skeletal structure of 5-(1-methylpropyl)nonane |
The longest chain of carbon atoms is clearly located at the bottom of the molecule. This chain gives it the root name nonane. The substituent is located at the second carbon atom. The substituent, however, is branched. If we were to name it alone, we would call it 1-methylpropane. But it is a substituent, so we must use the suffix -yl. The molecule is named 5-(1-methylpropyl)nonane.
Alkene nomenclature
Alkenes are hydorcarbons containing one or more double bonds. Alkenes are named using the same general naming rules for alkanes, except that the suffix is now -ene. There are a few other small differences:
- The main chain of carbon atoms must contain both carbons in the double bond. The main chain is numbered so that the double bond gets the smallest number.
- Before the root name, the number of the carbon atom at which the double bond starts (the smaller number) is written.
- If more than one double bond is present, the prefixes di-, tri-, tetra-, etc. are used before the -ene, and (strangely) the letter "a" is added after the prefix for the number of carbon atoms.
Alkyne nomenclature
Hydorcarbons containing one or more triple bonds are called Alkynes. Alkynes are named using the same general procedure used for alkenes, replacing the suffix with -yne. If a molecule contains both a double and a triple bond, the carbon chain is numbered so that the first multiple bond gets a lower number. If both bonds can be assigned the same number, the double bond takes precedence. The molecule is then named "n-ene-n-yne", with the double bond root name preceeding the triple bond root name (e.g. 2-hepten-4-yne).
| Previous topic: Basic bonding concepts |
2. Naming hydrocarons
Back to index |
Next topic: Naming functional groups |